Chemistry of Poisonous Plants
Hopewell Valley Student Podcasting Network
Chemistry Connections
Chemistry of Poisonous Plants
Episode #15
Welcome to Chemistry Connections, my name is Brian Chin and I am your host for episode 15 called Chemistry of Poisonous Plants. My name is Rey Riordan and I am also your host for this episode. Today we will be discussing various poisonous plants and how chemistry explains why they’re so dangerous.
Segment 1: Introduction to Chemistry of Poisonous Plants
Most people instantly think of poison ivy when they think of poisonous plants. The familiar itchiness, irritation, red skin. However, there are many other poisonous plants out there that are much more deadly. “Poisonous plants” are formally defined as plants that when touched or eaten in sufficient quantities are harmful or even fatal to organisms.
So, let’s talk about some examples, starting with the water hemlock:
- Water hemlock has many nicknames: beaver poison, devil’s flower, break-your-mother’s-heart
- According to Christianity, the water hemlock, which is native to the Mediterranean region, became poisonous after growing on the hillside of Jesus’ crucifixion
- On the outside, the water hemlock also gives signs of its poisonous nature. Its stem is streaked with purple and red and the leaves release an odor when crushed
- This plant famously killed the philosopher Socrates after he drank hemlock tea – Socrates felt numbing sensation that spread throughout body before he died
The stinging nettle is another extremely dangerous plant:
- The nettle, which can grow up to 7 feet tall, has stinging hairs known as trichomes on its green leaves (can be as big as 6 inches) and stem
- These trichomes inject harmful chemicals upon contact
- Because of its unique effects, the nettle has even impacted Western culture to a certain extent
- Aesop had a fable called “The Boy and the Nettle”
- The English word “nettled,” which denotes someone who’s irritated, is also derived from the properties of the stinging nettle
Segment 2: The Chemistry Behind Poisonous Plants
- Poison ivy
- Oxidation of urushiol in body
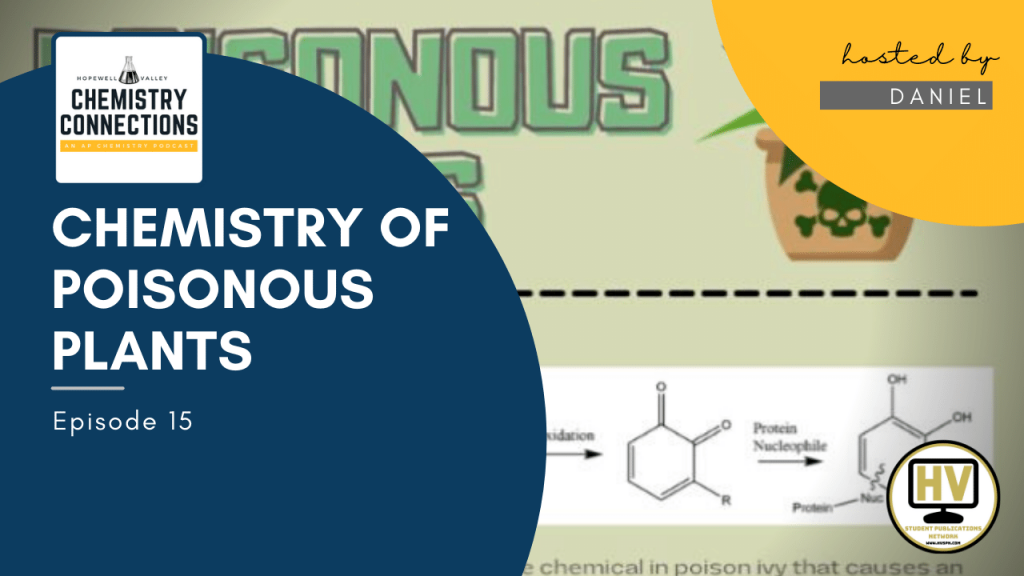
- Urushiol is the chemical in poison ivy that causes the allergic reaction. It’s a type of molecule known as a catechol, which means that it has a ring of six carbon atoms with alcohol (OH) groups attached to two of them, and then a string of trailing hydrocarbons (as shown in diagram).
- When something brushes up against poison ivy and urushiol comes into contact with air as a result, it reacts with the O2 molecules in the air and becomes oxidized. The H atoms are broken off, which means that an electron is lost and the oxidation number of O increases from -2 to -1 to compensate (this is what oxidation is).
- Oxidized urushiol with two double-bonded oxygens is then able to react with and stick to certain proteins of the skin. When reacted with a protein, urushiol acts as a hapten, which means that it causes an immune system response by changing the shape of the protein and making it seem foreign and dangerous to the body. This is what actually causes the allergic reaction of rashes and blisters that poison ivy is so well-known for.
- Water hemlock
- Cicutoxin
- Cicutoxin often more concentrated in hemlock’s roots – so don’t touch roots
- Cicutoxin’s chemical formula is C17H22O2
- Qualifies as alcohol because two hydroxyl groups (OHs) attached to carbon atoms that are part of a larger hydrocarbon chain
- Chemically, cicutoxin causes neuronal depolarization – essentially, the electric charge in a neuron cell changes so inside of cell becomes less negative than outside
- Too much neuronal depolarization causes cells to become overactive – overactive cells is the reason why cicutoxin damages nervous system and causes seizures – if seizures aren’t treated, can lead to swelling in brain, muscle breakdown, blood becoming too acidic
- Other symptoms include nausea, vomiting, abdominal pain, tremors
- Mass spectrometry
- Hospital labs use mass spectrums to see whether or not patient’s blood has cicutoxin
- After substance put in mass spectrometer, mass spectrum is produced. Mass spectrums essentially show peaks – each peak represents an atom or atoms (which can be identified using the mass) – height of peak shows abundance of atom/atoms
- When put in mass spectrum, cicutoxin always shows up the same way (kinda like person’s fingerprint) – same peaks when cicutoxin breaks up into smaller parts – doctors can thus easily identify cicutoxin even if other substances in blood
- Stinging nettle
- Neurotransmitters: histamine, acetylcholine, serotonin
- To cause pain, the stinging nettle mainly injects neurotransmitters such as histamine, acetylcholine, and serotonin. Neurotransmitters usually function as chemical messengers in the body by carrying chemical signals, but in this case they function as irritants and cause a painful reaction.
- For example, histamine normally responds to allergies and causes inflammation in the affected area, allowing the immune system to do its repair work. However, when injected unnecessarily, it results in unwanted inflammation and pain.
- Acids: formic acid, tartaric acid, oxalic acid (low concentration of formic acid)
- The stinging nettle also injects a number of acids, which are thought to either cause pain or extend the pain duration.
- Although present in a low concentration in stinging nettles, formic acid is capable of causing a stinging sensation and is present in many poisons such as ant venom. It has a chemical formula of HCOOH, and is an acid because it dissociates in water to form hydronium ions and its conjugate base HCOO-. It has an acid dissociation constant of 1.8 x 10^-4, which refers to the ratio between the products and reactants at equilibrium during its dissociation. With a small dissociation constant, formic acid is a weak acid, meaning that very little of it dissociates in water.
Segment 3: Personal Connections
Rey: The topic popped into my head because my father gets hit by poison ivy very frequently after yard work. He constantly complains about the itch and the fact that there is no particularly effective way to treat it. This got me wondering why this was so, and what chemicals were involved in the reaction. In addition, I’ve never gotten a poison ivy rash even while doing similar yard work. Thus, I also wondered whether it was possible to be immune to the reaction-causing chemicals in poison ivy.
Brian: I became interested in this topic after researching more about poisonous plants. It absolutely boggled my mind that some of these tiny, seemingly harmless looking plants could do so much damage on human beings. After finding out that these plants could even cause death, I wanted to research how this could be so. In my opinion, this topic is important because people should be aware that poisonous plants exist and they should be careful when doing things like hiking.
Thank you for listening to this episode of Chemistry Connections. For more student-ran podcasts and digital content, make sure that you visit www.hvspn.com.
Sources:
- Types of poisonous plants
- Poison Ivy (CDC)
- Intro to plant poisons (Brit)
- Definition of poisonous plant
- Water hemlock interesting background
- Poison Ivy reaction explanation
- Stinging nettle overview
- Stinging Nettle chemistry
- Water hemlock chemistry
- Poison Ivy (Wiki)
- More poison ivy
- Histamines
- Formic acid
Music Credits
Warm Nights by @LakeyInspired

